Scheelite
Scheelite
CaWO₄ Properties
- Category
- Mineral
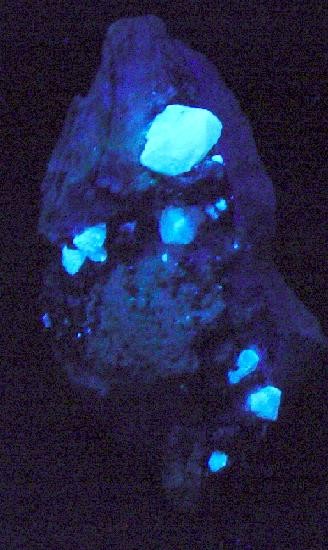
Scheelite is a calcium tungstate mineral (CaWO₄) that represents one of the world's principal industrial sources of tungsten. It crystallizes in the tetragonal crystal system and is easily recognized by its characteristic blue-violet fluorescence under ultraviolet light, a property so distinctive that the mineral is frequently employed as an indicator in mineral prospecting.
Scheelite is a calcium tungstate mineral that plays a crucial role in global mining industry as the primary source of tungsten, an essential element for high-strength alloys, filaments for incandescent bulbs, and chemical catalysts. Its name honors Swedish chemist Carl Wilhelm Scheele, who in 1781 identified tungstic acid in this mineral. It crystallizes in the tetragonal crystal system with a characteristic pseudo-octahedral crystal habit, often displaying well-developed pyramidal faces. Its hardness of 4.5–5 on the Mohs scale makes it relatively friable, but its high density (approximately 6.1 g/cm³) facilitates separation by gravity during concentration.
Scheelite typically forms in hydrothermal environments associated with granitic intrusions, where tungsten- and calcium-rich fluids interact with host rocks. The most common mineralogical associations include fluorite, molybdenite, pyrite, cassiterite, and quartz. The most important deposits are found in China (which supplies approximately 60% of global production), followed by Russia, Canada, Austria, and Peru. The most fascinating property of scheelite is its intense fluorescence under short-wave ultraviolet radiation, which produces blue-violet luminescence due to the presence of tungsten ions and crystal defects. This characteristic is not only a fundamental diagnostic tool for the mineralogist, but also represents a practical prospecting method: tungsten prospectors use portable ultraviolet lamps to locate scheelite in rock outcrops.
Scheelite crystallizes in the tetragonal crystal system, space group I41/a, with lattice parameters a ≈ 5.24 Å and c ≈ 11.38 Å. The structure is isomorphous with powellite (MoWO₄) and exhibits tetrahedral coordination of tungsten by oxygen atoms, while calcium shows octahedral coordination. The calculated density is approximately 6.12 g/cm³; hardness is 4.5–5 Mohs. The ordinary refractive index (ω) is approximately 1.920 and the extraordinary refractive index (ε) is approximately 1.936, with positive birefringence of approximately 0.016. Scheelite is anisotropic and exhibits weak pleochroism.
Fluorescence is the most characteristic diagnostic property: under short-wave ultraviolet radiation (254 nm) it emits intense blue-violet fluorescence, while under long-wave (365 nm) the fluorescence is generally weaker. This luminescence is attributed to electronic transitions of tungsten ions and defects in the crystal structure. Raman spectroscopy shows characteristic bands around 900 cm⁻¹ (W–O stretching) and 400–500 cm⁻¹ (bending). Under transmitted light microscopy, scheelite appears colorless to pale yellow; under reflected light it shows moderate reflectivity. The chemical composition is very pure in natural specimens, although traces of molybdenum may substitute for tungsten, forming a solid solution series with powellite. Solubility in strong acids is low; scheelite is insoluble in dilute hydrochloric acid at room temperature.
Mining localities
- Cina (Hunan, Guangdong, Yunnan)
- Russia (Siberia orientale)
- Canada (British Columbia)
- Austria (Salzburg)
- Perù (Andes)
- Portogallo (Beiras)
- Kazakistan
- Tailandia