Aragonite
Aragonite
CaCO₃ Properties
- Category
- Mineral
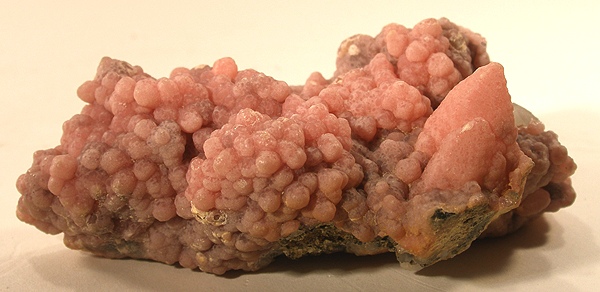
Aragonite is a calcium carbonate (CaCO₃) that crystallizes in delicate, needle-like forms, often grouped in flower-like or star-shaped structures. It is the most unstable mineral among the polymorphs of calcium carbonate and slowly transforms into calcite over millions of years.
Aragonite represents one of three principal polymorphs of calcium carbonate, alongside calcite and vaterite. Unlike calcite, which crystallizes in the trigonal crystal system, aragonite adopts the orthorhombic crystal system, with space group *Pmcn*. This different crystal structure renders it thermodynamically unstable at ordinary temperatures and pressures: over geological time, aragonite gradually transforms into calcite, the most stable polymorph. It forms characteristic acicular crystals (thin needles) or pseudohexagonal habits, often grouped in radiated aggregates reminiscent of cactus flowers. It is commonly found in coastal sedimentary environments, in mollusk shells, coral skeletons, and shallow marine sediments. It is also an important component of nacre (mother-of-pearl) and forms in cave deposits of stalactites and stalagmites where waters are rich in calcium. Its hardness of 3.5–4 on the Mohs scale makes it fragile and easily soluble in dilute acids.
Crystal system: orthorhombic, space group *Pmcn* (Z = 4). Lattice parameters: a = 5.741 Å, b = 7.975 Å, c = 4.959 Å. Density: 2.93–2.95 g/cm³. Mohs hardness: 3.5–4. Cleavage: perfect along (10), good along (110). Crystal habit: acicular, pseudohexagonal, often in radiated or fibrous aggregates. Color: colorless, white, yellowish, pink, brown; transparent to translucent. Refractive index: α = 1.530, β = 1.682, γ = 1.685; positive birefringent. Pleochroism: weak. Solubility: vigorous effervescence in dilute HCl (10%). IR spectroscopy: characteristic carbonate absorptions at ~1500 cm⁻¹ and ~875 cm⁻¹. UV fluorescence: generally inert, occasionally weakly fluorescent (blue or orange). Thermal stability: progressive transformation toward calcite at ordinary temperatures; inversion is accelerated by pressure and the presence of impurities. Mineral associations: calcite, gypsum, halite, celestine, barite; frequent in marine sediments, shells, corals, pearls. Genesis: direct precipitation from seawater, biogenic formation (skeletons), diagenesis of coastal sediments, cave deposits.
Mining localities
- Molini di Trieste (Friuli-Venezia Giulia, Italia) — cristalli pseudoesagonali in sedimenti marini
- Grotta di Castellana (Puglia, Italia) — aragonite in stalattiti e stalagmiti
- Flos Ferri, Sardegna (Italia) — forme aciculari in cavità
- Spagna — giacimenti costieri e depositi di grotta
- Messico — aragonite biogenica in conchiglie e sedimenti marini
- Giappone — forme pseudoesagonali in ambienti marini
- Namibia — aragonite in depositi sedimentari costieri
- Marocco — cristalli aciculari in cavità
- Perù — aragonite in sedimenti marini e depositi di grotta